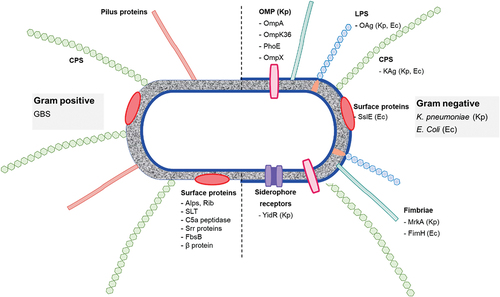
Neutron macromolecular crystallography (NMX) is a unique method for locating hydrogen atoms. Its effectiveness is greatly improved by using deuterium-labeled molecules. Human glycans are the targets of virulence factors from pathogens, such as soluble lectins from the opportunistic bacterium Pseudomonas aeruginosa. Deuterated galactose was produced by hydrogen isotope exchange and co-crystallized with a fully deuterated bacterial receptor, LecA, a lectin involved in P. aeruginosa tissue adhesion and biofilm formation. The structure of the complex determined through neutron diffraction shows the positions of all hydrogen atoms as deuterium, emphasizing the role of a charged histidine in the binding site, the bridging water molecule, and the impact of the coordinating calcium ion on nearby hydrogen bonds. LecA is a target for pathoblockers, and these structural details can aid in the design of glycomimetics to combat multidrug-resistant infections.
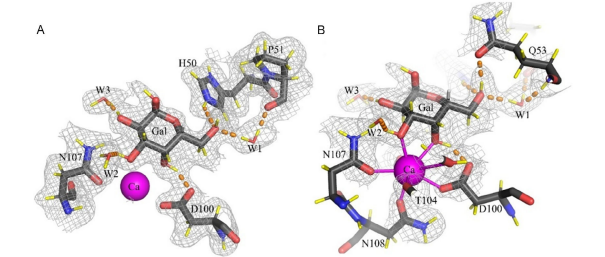
Figure. The galactose-binding site in the neutron structure of the D-LecA/Gal-d10 complex (chain H). The hydrogen bonds are shown as orange dashed lines. The 2mFo–DFc neutron density map (gray mesh) is contoured at 0.8σ. (A) Upper view of the binding site with Q53 omitted for clarity. (B) Alternative view including Q53 and displaying the coordination of the calcium ion.