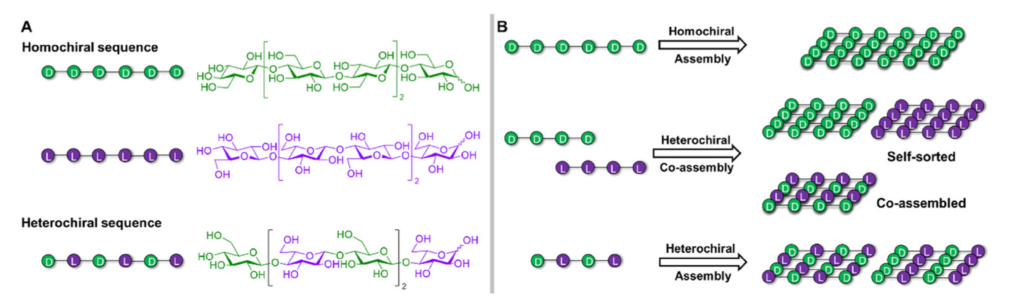
Chirality has become a key design principle for the construction of peptide materials. In contrast, the systematic use of chirality to develop glycan materials remains largely unexplored, despite the rich chiral diversity of carbohydrates, with both D- and L-configurations readily found in nature. Here, we highlight the added value of exploring mirror-image glycans to customize carbohydrate materials. By examining the unique chiral features of carbohydrates in comparison to peptides, the authors demonstrate how these characteristics provide valuable opportunities to influence and better understand the rules that control glycan assembly. The authors explore examples of carbohydrate materials based on individual enantiomers, the co-assembly of racemic mixtures, and the formation of heterochiral sequences. Each section starts with key insights from peptide materials, serving as inspiration and guidance for future glycan assembly designs.