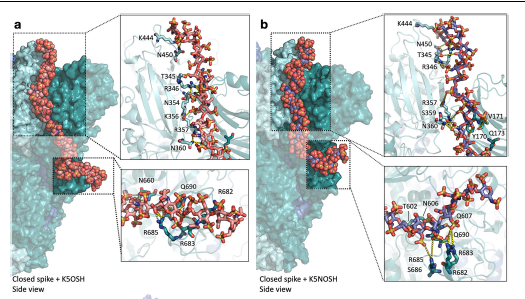
PSARS-CoV-2 spike glycoprotein is a promising drug target due to its crucial role in viral infection.Heparin, a long linear polysaccharide that inhibits SARS-CoV-2 infection by acting on spike, has limited antiviral applications due to its anticoagulant effect. E. coli K5 polysaccharides share the same structure as the heparin precursor and can be chemically modified to be devoid of anticoagulant activity. Here, biochemical assays and computer simulations reveal that K5 with high degree of sulfation at O- (K5OSH) or N- and O-positions (K5NOSH) bind spike with higher affinity than heparin, preventing its binding to ACE2 and furin cleavage. This mechanism is supported by cell syncytia assays showing that K5OSH and K5NOSH inhibit viral infection by blocking membrane fusion. Infection assays for SARS-CoV-2 Wuhan-Hu-1 and Omicron BA.1 variants corroborate their antiviral activity. These results support the therapeutic potential ofK5OSHandK5NOSHagainstSARS-CoV-2, with K5OSH displaying more promising activity profile.riming