The biosynthesis of the ABO and all its related precursors such as H, I and P blood group system glycolipids (glycosphingolipids) all originate with glucose (Glcβ1) linked to a ceramide (Figure 5).
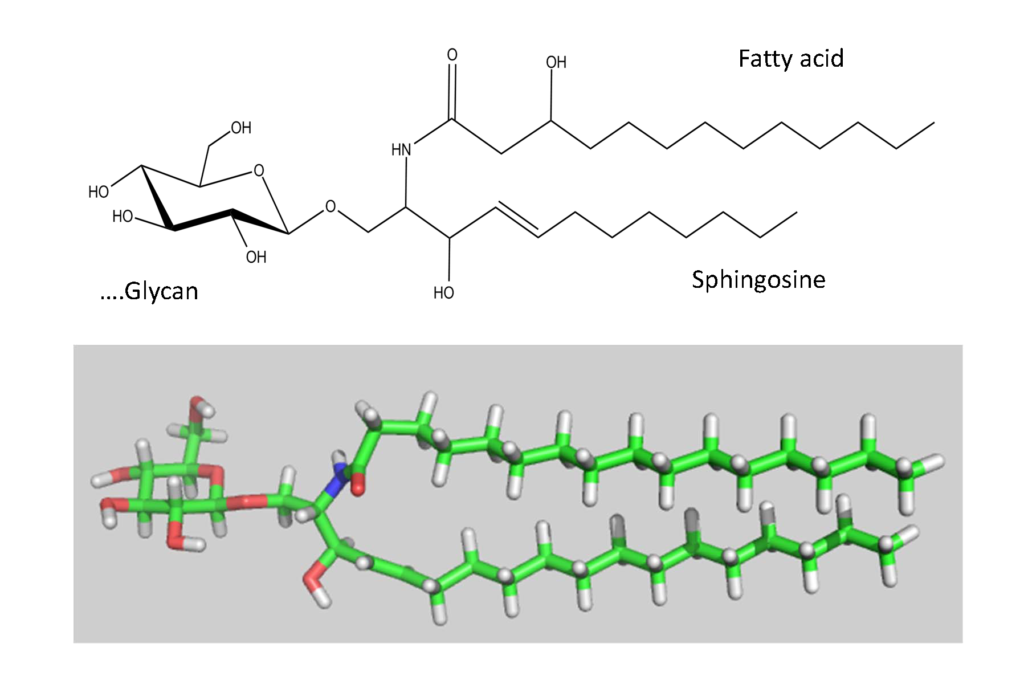
A galactose residue in β4 linkage (Galβ4) forms lactosylceramide (Galβ4Glcβ1-Cer) (Figure 6).
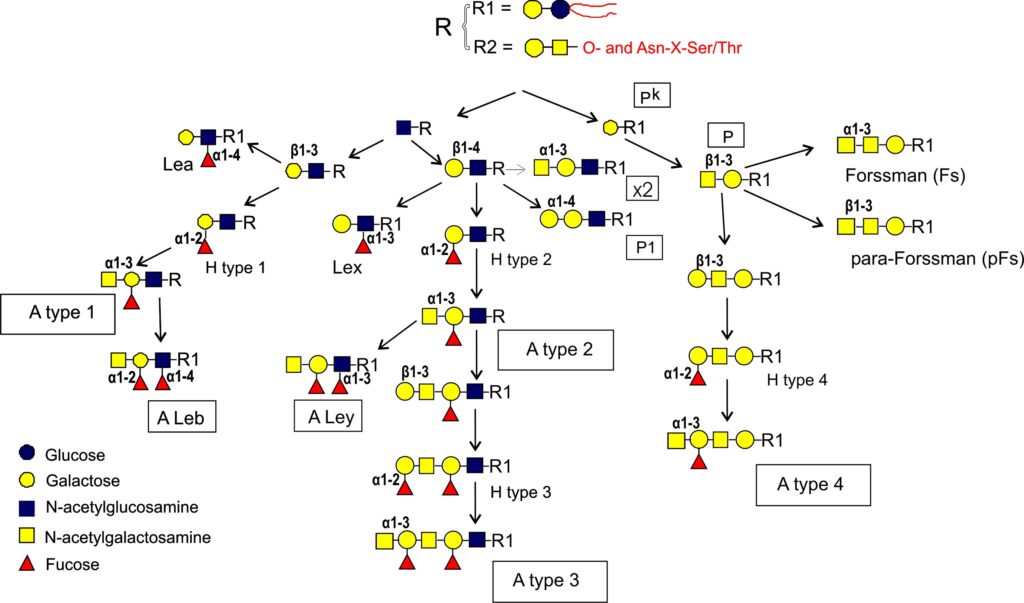
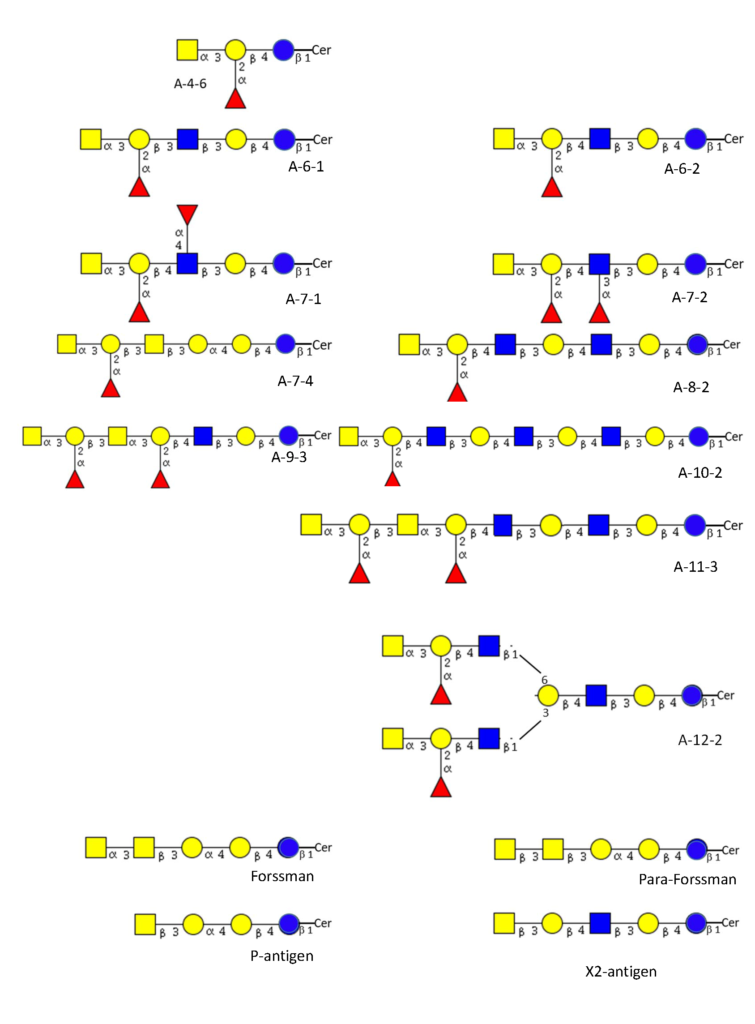
From this precursor two alternative pathways exist, one via lactotriaosylceramide towards type 1, 2, 3 precursors Rege et al., 1963Donald,1981Clausen et al., 1986Bremer et al., 1986, and the other via globotriaosylceramide (Gb3) towards type 4 precursor Bremer et al., 1984. In more detail, after lactotriaosylceramide (GlcNAcβ3Galβ4Glcβ1Cer) is formed and biosynthesis splits into two alternative pathways : the addition of Gal with a β3 linkage leads to lactotetraosylceramide (Lc-4 or type 1 precursor, Galβ3GlcNAcβ3Galβ4Glcβ1Cer) Reget et al., 1963, or Gal in β4 linkage leads to neolactotetraosylceramide (nLc-4 or type 2 precursor, Galβ4GlcNAcβ3Galβ4Glcβ1Cer) Reget et al., 1963. Both of these precursors can be fucosylated by the fucosyltransferase 2 (Fuc-TII) also known as the Se enzyme to form H type 1 and H type 2 antigens while the fucosyltransferase 1 (Fuc-TI) also known as the H enzyme can only modify the type 2 precursor to form H type 2. The H type 2 antigen may be extended by the action of GTA and GTB. The synthesis by GTA will be described in this section and in the section of the synthesis of glycoproteins. GTA adds the α3 GalNAc (α3 Gal) residue and thus forming A type 2 antigen Holgersson et al., 1992Hakomori et al., 1972 which may be further extended with a Fucα2Galβ3 disaccharide forming the H type 3 (Fucα2Galβ3GalNAcα3-R). This may also be further modified into A type 3 (“repetitive A” or A type 3) (GalNAcα3[Fucα2]Galβ3GalNAcα3-R) Clausen et al., 1985 (Figure 4).
The biosynthesis of the type 4 pathway also originates from the lactosylceramide precursor where a Gal residue is added in an α4 linkage leading to globotriosylceramide (Gb3, also known as Pk) and then by adding a β3GalNAc to globotetraosylceramide, Gb4 (globoside or also known as the P antigen Kannagi et al., 1984 (Figure 4). From here three alternative pathways exist where Gb4 may be further extended into other structures. Elongation with the H-disaccharide forms H type 4 (globo-H) Kannagi et al., 1984Clausen et al., 1984 which can be further modified by the A1 transferase into A type 4 (A-7-4, globo-A) Clausen et al., 1984. The A2 transferase appears not able to convert H type 4 precursor into A-7-4 or at best very ineffectively Clausen et al., 1984Svensson et al., 2009. The other two pathways do not result in blood group A antigens. The addition of GalNAc with β3 linkage creates the p-Fs (para-Forssman) glycolipid Ando et al., 1982 while the addition of GalNAc with α3 linkage creates the Fs (Forssman) glycolipid Siddiquiet al., 1972Haslam & Baenziger, 1996Tamakawa et al., 1996. p-Fs is expressed in humans and Fs is widely seen as an animal antigen although a few publications have indicated its expression in human tissue, especially in tumors Hakomori et al., 1977Hakomori, 1984Breimer, 1985 and recently it has been established to be present on some human red cells as the blood group system, FORS Svensson et al., 2012. The antigens p-Fs and Fs (FORS1) although not blood group A antigens can react with some anti-A reagents Svensson et al., 2012. Figure 7 shows structurally characterized blood group A glycolipid structures known in red cell membranes and non-blood group A glycolipids that may cross-react with anti-A reagents.