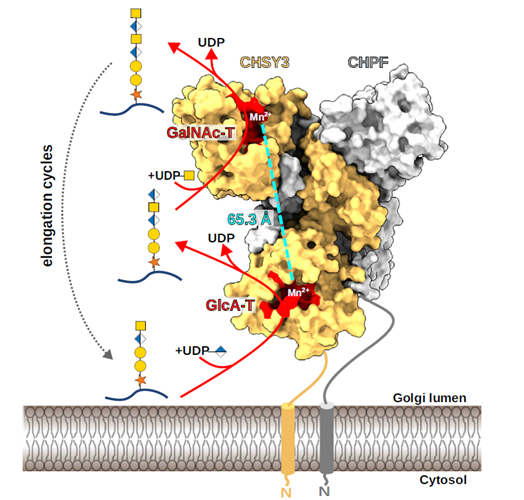
Chondroitin sulfates are complex polysaccharides that regulate diverse biological processes at the cell surface and within the extracellular matrix. In this study, the authors identify four heterodimeric enzyme complexes that mediate chondroitin sulfate chain polymerization in humans: CHSY1–CHPF, CHSY1–CHPF2, CHSY3–CHPF, and CHSY3–CHPF2. Using a custom in vitro glycosylation assay using chemoenzymatically synthesized fluorescent substrates, the authors show that all four complexes can polymerize chondroitin sulfate chains. A cryo-electron microscopy structure of the CHSY3–CHPF complex reveals the molecular basis of the polymerization reaction. Structural analysis indicates that CHSY1 and CHSY3 harbor the catalytic activities, whereas CHPF and CHPF2 primarily serve a stabilizing function within the complexes. Mutational studies of purified complexes, together with in cellulo complementation assays, confirm that only CHSY1 and CHSY3 possess bifunctional glycosyltransferase activity. Finally, the spatial organization of the catalytic sites supports a non-processive, distributive mechanism for chondroitin sulfate chain polymerization.