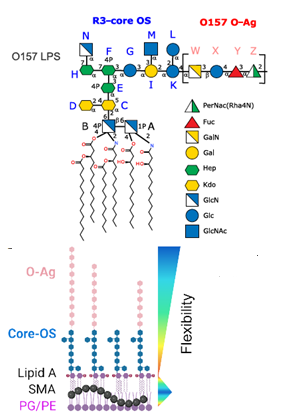
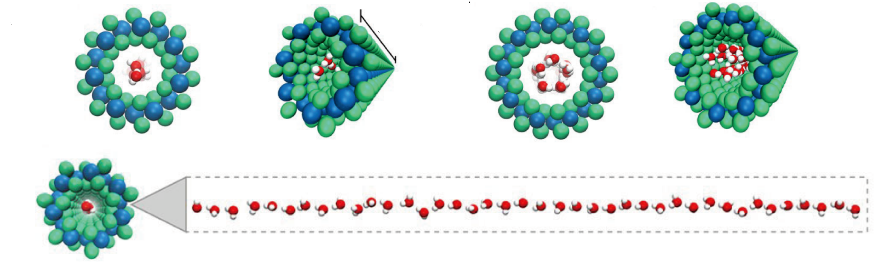
Lipopolysaccharides (LPSs) are essential components of the outer membranes of gram-negative bacteria, crucial for antimicrobial resistance, virulence, and triggering the host immune response. Self-assembled particles displaying LPSs are important for biophysical studies of bacterial surface behavior under biomimetic conditions. Using styrene-maleic acid copolymers, LPS nanoparticles were generated from either extracted LPS or directly from purified outer membranes. These nanoparticles, originating from pathogenic O157:H7 or laboratory E. coli strains, are size-defined and yield high-resolution nuclear magnetic resonance spectra. They have been effectively used to study molecular recognition by a human C-type lectin receptor of the immune system and to analyze interactions with polymyxin antibiotics through various biophysical techniques. This research highlights the significance of LPS nanoparticles as bacterial surface mimetics and opens new possibilities for exploring LPS structure and interactions. The ability to produce well-characterized LPS nanoparticles offers a valuable tool for understanding the molecular mechanisms of bacterial pathogenicity and immune responses.