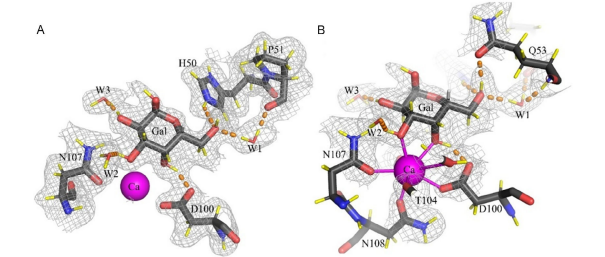
Bacterial capsules play a critical role in host-pathogen interactions. They provide a protective envelope against host recognition, leading to immune evasion and bacterial survival. Here, we define the capsule biosynthetic pathway of Haemophilus influenzae serotype b (Hib), a Gram-negative bacterium that causes severe infections in infants and children. Reconstitution of this pathway allowed fermentation-free production of Hib vaccine antigens from widely available precursors and detailed characterization of the enzymatic machinery. The X-ray crystal structure of capsule polymerase Bcs3 reveals a multi-enzyme machine that adopts a basket-like shape, providing a protected environment for the synthesis of the complex Hib polymer.
This architecture is commonly used for surface glycan synthesis by both Gram-negative and Gram-positive pathogens. Supported by biochemical studies and comprehensive 2D nuclear magnetic resonance, our data explain how the ribofuranosyltransferase CriT, the phosphatase CrpP, the ribitol phosphate transferase CroT and a polymer-binding domain function as a unique multi-enzyme assembly.