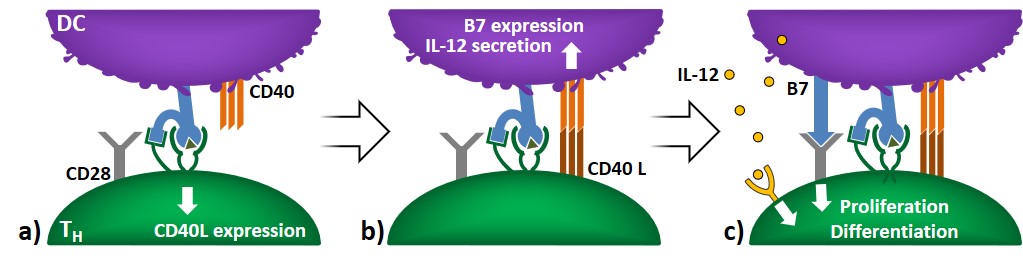
TH cell activation and licensing
The first event in helper T-cell activation involves DC-mediated antigen recognition (with or without B7), which causes the expression of co-stimulatory CD40L on TH cells (Figure 7a). The binding of CD40L to CD40 increases the expression of B7 in DCs and stimulates cytokine secretion (e.g. IL-12). This causes the activation of TH proliferation and differentiation, and increases the expression profile of co-stimulatory B7, improving DC’s antigen presentation (Figure 7b-c). The phenomenon whereby activated T lymphocytes make APCs more powerful stimulators of immune responses is referred to as licensing.
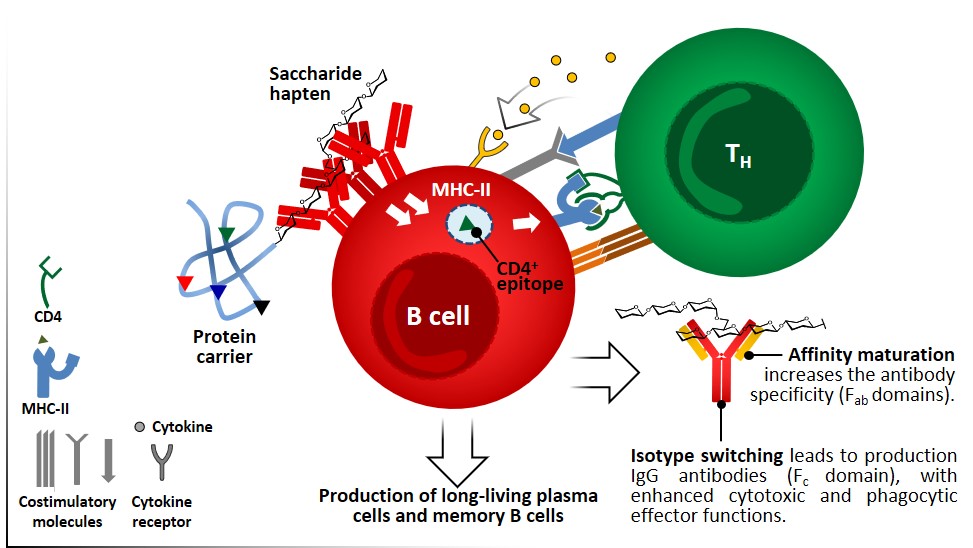
B-cell activation
Antigens are presented generally in its intact form to naïve B cells through BCRs, as mentioned before this represents a fundamental distinction between B and T cells. Antigen-mediated crosslinking of BCRs by T-cell independent antigens like polysaccharides and glycolipids, which normally display multiple identic epitopes, induces events such as proliferation, increased expression of costimulatory molecules and cytokine receptors (Figure 4). Proteic antigens, once internalized through BCRs, are processed inside the B cell and presented upon MHC II molecules. In the lymph nodes, DC-activated CD4+ TH cells that express CD40L (Figure 7) can recognize the same antigen on MHC II of B cells and strongly activate their proliferation and differentiation in the germinal center, producing long-living Ab-secreting plasma cells and resting memory cells (Figure 9). B cell activation by TH cells also triggers isotype switching, by which IgM antibodies are replaced by the more effective IgG isotype, and affinity maturation, leading to a more specific population of antibodies.
Therefore, T-dependent B-cells responses exploit at least two kinds of epitopes: (i) a surface epitope of variable nature, which is recognized and internalized through BCRs; (ii) an internal linear peptide which undergoes the MHC II pathway and is recognized by effector CD4+ cells. The antibodies that are produced are specific for the surface epitopes recognized by BCRs, leading to a fine humoral response which is independent of the fact that TH cells recognize restricted epitopes (linear epitopes of processed peptides). This principle is known as hapten-carrier effect and represents the basis for the development of conjugate vaccines: carbohydrate epitopes conjugated to proteins are recognized by BCRs, internalized and peptide epitopes resulting from the carrier protein are exposed through MHC II complexes, thus enabling TH cells to activate B cells, and to stimulate the production of high-affinity IgG Abs against the carbohydrate epitope itself. To stimulate an effective response, the hapten (e.g. saccharide epitope) and the carrier portion must be covalently linked rather than administered separately. The B-TH cells interaction is class II MHC restricted, so activated TH cells can only cooperate with B cells that present the same restricted antigen which was involved in the activation of naïve T cells.