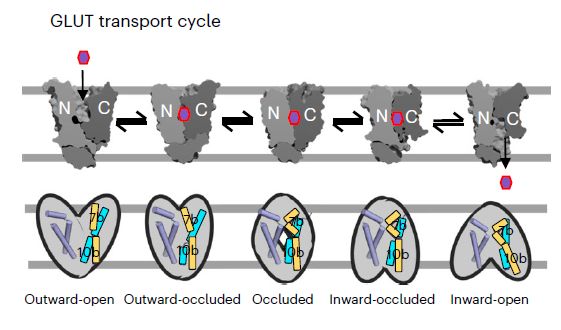
Sugar porters belonging to the major facilitator superfamily (MFS) represent one of the largest groups of transporters across all kingdoms of life. In mammals, glucose transporters (GLUTs) mediate organism-wide sugar distribution, yet the molecular basis of substrate specificity remains unclear. The bacterial xylose transporter XylE serves as a model for GLUTs. Although xylose and glucose bind with similar affinity, xylose is transported, whereas glucose acts as an inhibitor. Using saturation transfer difference (STD) nuclear magnetic resonance (NMR) spectroscopy, the authors distinguished transported sugars from sugar inhibitors. Their findings revealed that only transported sugars generate STD NMR signals, which are abolished for xylose when the transporter XylE is trapped in either outward- or inward-facing conformations. Engineering the sugar-binding pocket and gating helix TM7b enabled glucose transport by XylE, as evidenced by the corresponding STD signals. Using complementary molecular dynamics simulations, together with structural, biochemical, and STD NMR analysis of related parasitic and mammalian GLUTs, the authors identified TM7b as a key determinant of occluded state formation. The authors conclude that, rather than the initial substrate-binding event observed in experimental structures, formation of a substrate-induced transition-state intermediate is the primary determinant of specificity in transporters.