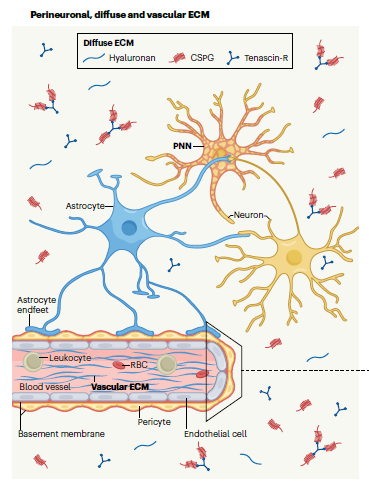
The six mammalian glycosaminoglycans (GAGs), chondroitin sulfate, dermatan sulfate, heparin, heparan sulfate, hyaluronan, and keratan sulfate, are linear polysaccharides. Except for hyaluronan, they are sulfated to various extent, and covalently attached to proteins to form proteoglycans. GAGs interact with growth factors, morphogens, chemokines, extracellular matrix proteins and their bioactive fragments, receptors, lipoproteins, and pathogens. These interactions mediate their functions, from embryonic development to extracellular matrix assembly and regulation of cell signalling in various physiological and pathological contexts such as angiogenesis, cancer, neurodegenerative diseases, and infections.
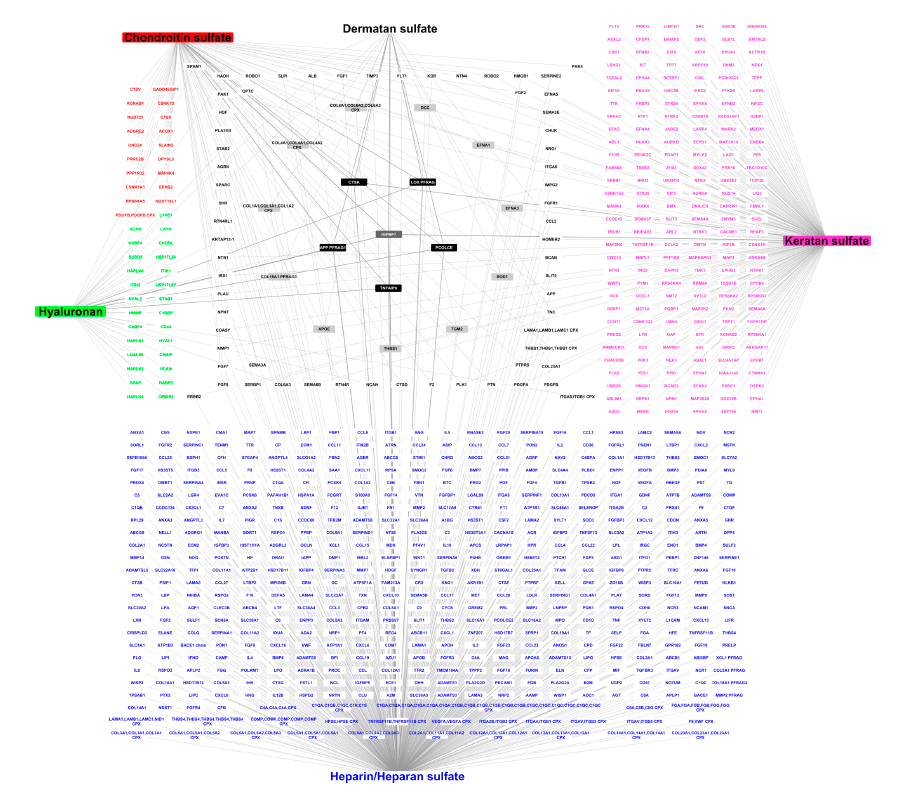
The authors provide an overview of GAG–protein interactions (i.e., specificity and chemical features of GAG- and protein-binding sequences), and review the available GAG–protein interaction networks. They also describe the first comprehensive draft of the GAG interactome composed of 832 biomolecules (827 proteins and five GAGs) and 932 protein–GAG interactions. This network is a scaffold, which in the future should integrate structures of GAG–protein complexes, quantitative data of the
abundance of GAGs in tissues to build tissue-specific interactomes, and GAG interactions with metal ions such as calcium, which plays a major role in the assembly of the extracellular matrix and its interactions with cells. Such a contextualized
interactome will be useful to identify druggable GAG–protein interactions for therapeutic purpose